“So far, we have not found anything that is majorly concerning; at most, we found slight body fever which then disappeared within two days,” he said during a virtual dialogue on 'Answering Doubts About Vaccines' on Tuesday.
He said 15 people have so far withdrawn from Phase 3 of the clinical trial, eight of whom withdrew due to unrelated illnesses and the remaining due to professional shifts.
"I think up until now the security can still be accounted for," he said, adding that Phase 3 of the clinical trial is not being carried out in a hurry.
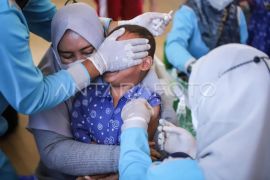
Rusmil said 1,620 volunteers have received the first dose of the vaccine, while 1,590 volunteers have been given two doses.
"The clinical trial process has been going on quite smoothly so far," he said.
As part of the trial, all volunteers who have received the COVID-19 vaccine shots will be monitored for six months.
Rusmil said the clinical trial is being carried out in a multi-center manner, as trials for the vaccine are being carried out in several countries, including Brazil and Turkey. The Indonesian government will compare the results of the Sinovac vaccine clinical trial in Bandung, West Java with those carried out in other countries.
"If the third phase is passed, the vaccines can be used and traded," he added.
Drawing from his own experience in clinical studies of vaccines, he said the clinical trial of the Sinovac vaccine so far has demonstrated its safety.
Of the many immunizations carried out in Indonesia, he continued, the possibility of a severe reaction, such as fainting after being vaccinated, is very small, with the incidence rate pegged at 0.1 to one in a million people receiving the shots. (INE)
Related news: Clinical trial of Sinovac COVID-19 vaccine ran smoothly: government
Related news: Indonesia to obtain 30 million COVID-19 vaccine doses at 2020-end
EDITED BY INE
Translator: Martha Herlinawati S/Aria Cind
Editor: Suharto
Copyright © ANTARA 2020