"DPR's Commission VI, Commission VIII, and Commission IX urge the minister of research and technology/head of BRIN (the National Research and Innovation Agency), minister of health, minister of industry, minister of BUMN (State Enterprises), head of BPPT (the Agency of Assessment and Application of Technology), head of LIPI (the Indonesian Institute of Sciences), head of LAPAN (the National Institute of Aeronautics and Space), and director of Eijkman Institute for Molecular Biology to immediately accelerate the mass production of COVID-19 detection devices," Sugeng Suparwoto, chairman of the House of Representatives' Commission VII, stated while announcing the recommendations of a joint meeting of the Commissions, here, Tuesday.
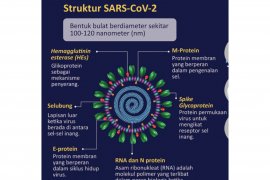
The COVID-19 equipment that must be produced on a massive scale comprise PCR test kits, RT Lamp, Turbidimetry and Colorimetric-based test kits, and non-PCR Rapid Diagnostic Test Kits (RDT), ventilators, mobile BSL-2 Laboratory, powered air-purifying respirators, and PPE.
The Parliamentarian Commissions also called on relevant ministries and institutions to comprehensively coordinate for an integrated and accelerated response to COVID-19.
They also appealed to the government to coordinate with those ministries and other relevant institutions in developing COVID-19 vaccines using virus isolates in Indonesia and expediting the implementation of alternative COVID-19 drug candidates, including quinine pill clinical trials; and studies of phytopharmaca herbs as immunomodulators, as well as serum, stem cells, and convalescent blood plasma therapy.
Related news: House of Representatives backs government regulation on COVID-19
Related news: APEC trade ministers issue statement on COVID-19 pandemic
The DPR encouraged the BUMN (SOE) minister and health minister to support state-owned hospitals, government hospitals, and private hospitals to purchase and use medical devices as the results of the COVID-19 Research and Innovation Consortium.
They also urged the research and technology minister/BRIN head, through the COVID-19 Consortium for Research and Innovation, to conduct research that can produce raw materials for medicines in the country to boost health resilience and sovereignty.
The Indonesian Parliament supports the Indonesian health minister to lower or halt the importation of medical devices in order to step up the use of Indonesia-made medical devices or that have been produced from the COVID-19 Research and Innovation Consortium Program.
The DPR has encouraged the health minister to ease regulations, including requirements and special protocols, for testing medical devices in order to speed up the use of medical devices and research and innovation products.
The DPR has called on the industry minister to support the COVID-19 Research and Innovation Consortium to work with partner industries.
During the meeting, Research and Technology Minister/Head of BRIN Bambang P. S. Brodjonegoro called upon the industry minister to help encourage industry partners to work together to produce innovations and technologies for handling the COVID-19 pandemic.
Brodjonegoro also appealed to the SOE minister to encourage SOE hospitals to use the COVID-19 Consortium's innovation and research products, including ventilators and COVID-19 detection devices.
Related news: Indonesians urged to be disciplined in practicing physical distancing
Related news: COVID-19 pandemic containable by June-July: spokesman
Translator: Fardah
Editor: Sri Haryati
Copyright © ANTARA 2020