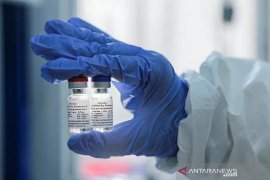
"Our hope is that by the end of 2021, our nation could produce the Red and White vaccine," said BPOM head Penny Kusumastuti Lukito during a workshop on the Red and White Vaccine here on Tuesday.
Of the six institutions developing the Red and White vaccine, two institutions have made the fastest progress — Airlangga University and the Eijkman Institute for Molecular Biology, she disclosed.
According to the BPOM, the Red and White vaccine being developed by the University of Airlangga (Unair) has now entered the preclinical or animal testing stage.
Unair is developing its Red and White vaccine candidate using an inactivated virus platform.
"From Airlangga University with the inactivated virus platform, it has started preclinical testing, animal testing has started as of yesterday (April 9, 2021). Alhamdullillah, it has started preclinical (trials)," Lukito said.
It is hoped that the pre-clinical trials and clinical trials for the vaccine being developed by Unair will be completed in October, 2021, so that production can begin by the end of 2021, she added.
Related news: Merah Putih vaccine to help Indonesia handle future pandemics: govt
For mass producing the vaccine, Unair has partnered with pharmaceutical company, PT Biotis, which is currently trying to obtain a Good Manufacturing Practice (GMP) certificate.
In the near future PT Biotis will receive the GMP certificate, which is likely in May, 2021, if all goes well, Lukito said.
Meanwhile, the vaccine seeds that are being developed by the Eijkman Institute for Molecular Biology will enter the preclinical stage soon, she added.
Eijkman is developing its Red and White vaccine candidate using a recombinant protein subunit platform.
It is hoped that the vaccine can enter the mass production stage in the third quarter of 2022, Lukito said.
The recombinant protein subunit platform is a newer vaccine development technology, she added.
Eijkman will partner with PT Bio Farma for mass production of its vaccine. "For Bio Farma, this is also something new, of course, concerning its production facilities," she said.
Four other institutions that are also developing Red and White vaccine candidates are the Indonesian Institute of Sciences (LIPI), the Gadjah Mada University (UGM), the University of Indonesia (UI), and the Bandung Institute of Technology (ITB).
Related news: Bio Farma to receive Eijkman's vaccine seed stock at March-end
Translator: Martha HS, Fardah
Editor: Rahmad Nasution
Copyright © ANTARA 2021