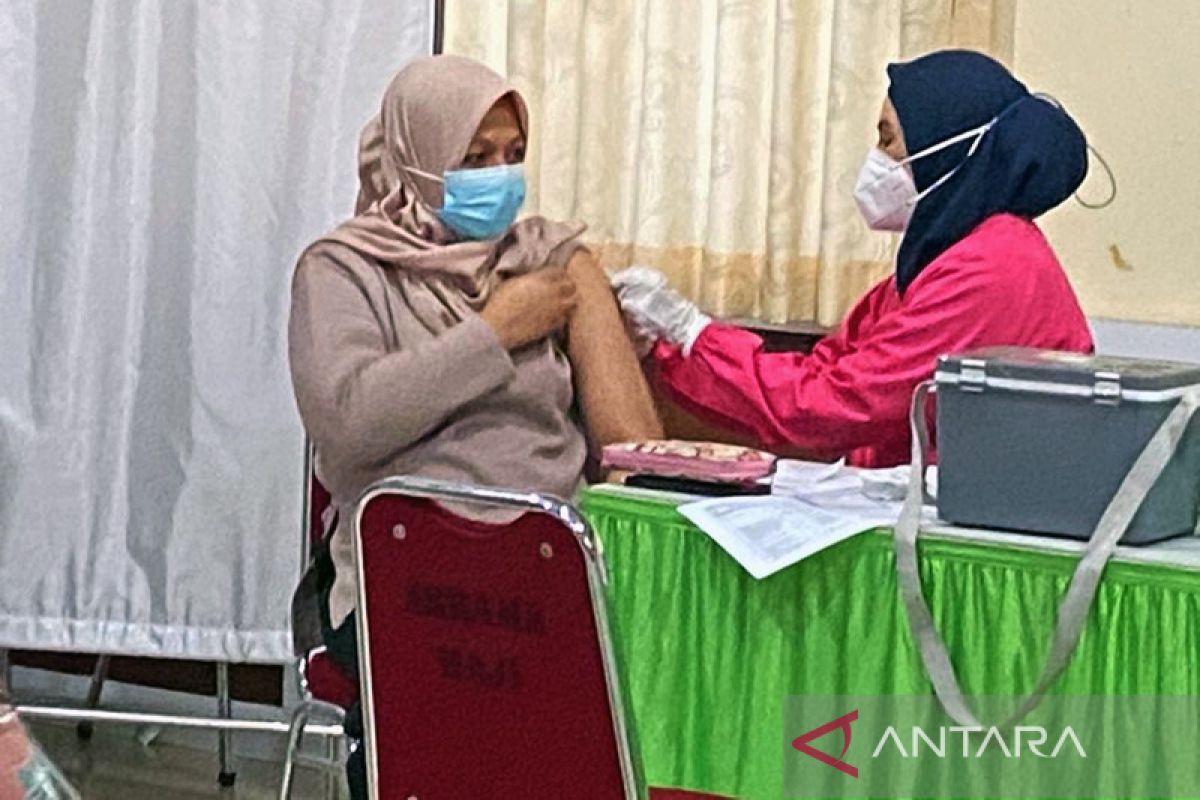
"If the production is sufficient and the compatibility factor with previous types of vaccines has met the requirements, then the import of vaccines should be replaced with domestic vaccines," he told ANTARA here Wednesday.
Listiyanto also viewed that the booster vaccine coverage, which is still low, is not considered to be too disruptive to economic recovery.
"The booster requirement is needed in line with the increase in positive cases of COVID-19 now, as economic recovery is important. However, controlling the pandemic is more important as a prerequisite for economic recovery, so I do not think that it will be too disturbing," he remarked.
He opined that currently, the threat of economic risks in terms of the energy and food price hike is graver than the health risks.
"Although this booster vaccine can work well, the government still has a duty to control the impact of energy and food prices that tend to rise," he pointed out.
He said that the national economic recovery program (PEN) should end this year, and next year, it should return to a fiscal discipline, with the APBN (State Budget) deficit expected to be lower than three percent of the GDP.
Earlier, Director of Bio Farma Honesti Basyir stated that after the phase-three clinical trial and having obtained an emergency use permit, or EUA, issued by BPOM at the end of July this year, the Merah Putih (Red and White) indigenous vaccine will be used for booster vaccine for adults and first- and second-dose vaccine for children.
Having the indigenous COVID-19 vaccine produced by Indonesia's state-owned company is a manifestation of the independence of the Indonesian nation during the pandemic, he affirmed.
Indonesia has, so far, been using imported COVID-19 vaccines, such as CoronoVac, Pfizer, Moderna, and AstraZeneca.
Related news: Phase 3 trial of Merah Putih vaccine on in Padang Pariaman
Related news: Merah Putih Vaccine phase 3 trial conducted alongside booster trial
Translator: Ilham Kausar, Fardah
Editor: Rahmad Nasution
Copyright © ANTARA 2022