#antibody
Collection of antibody news, found 167 news.
Celltrion Group announced today that the European Commission (EC) has approved Regkirona (regdanvimab, CT-P59), one of ...
Celltrion Group announced today that the European Medicine’s Agency’s (EMA) Committee for Medicinal Products for ...
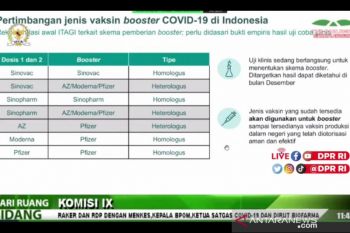
Allocation of the COVID-19 booster vaccine for citizens should take into account the state of global vaccination ...
News Focus
The administration of Joko Widodo (Jokowi) and Ma'ruf Amin has left no stone unturned in tackling the COVID-19 ...
Spokesperson of the COVID-19 Handling Task Force Prof. Wiku Adisasmito stressed that COVID-19 treatment drugs must ...
Celltrion Group announced that the Korean Ministry of Food and Drug Safety (MFDS) approved regdanvimab (CT-P59), a ...
RubrYc Therapeutics, Inc. (“RubrYc”) and iBio, Inc. (“iBio”, NYSEA: IBIO) announced today the closing of ...
The Jakarta Health Office has appealed to breastfeeding women to get vaccinated to reduce the risk of COVID-19 ...
Mothers who test positive for COVID-19 can breastfeed their babies, a Health Ministry nutritionist has said at an event ...
Dissemination of information pertaining to the benefits and importance of convalescent plasma donation should be ...
Identifying asymptomatic COVID-19 patients and monitoring the spread of the virus in the environment is difficult, ...
An epidemiologist at the Faculty of Public Health, University of Indonesia (FKM-UI) Pandu Riono, at a discussion ...
The West Java provincial government has begun offering medicine and vitamin assistance for COVID-19 patients conducting ...
Families have an important role to play as a bulwark against the spread of coronavirus infections, Minister of Home ...
Amid rising concerns over the threat and economic impact of COVID-19, Holista Colltech Ltd ("Holista") has been ...