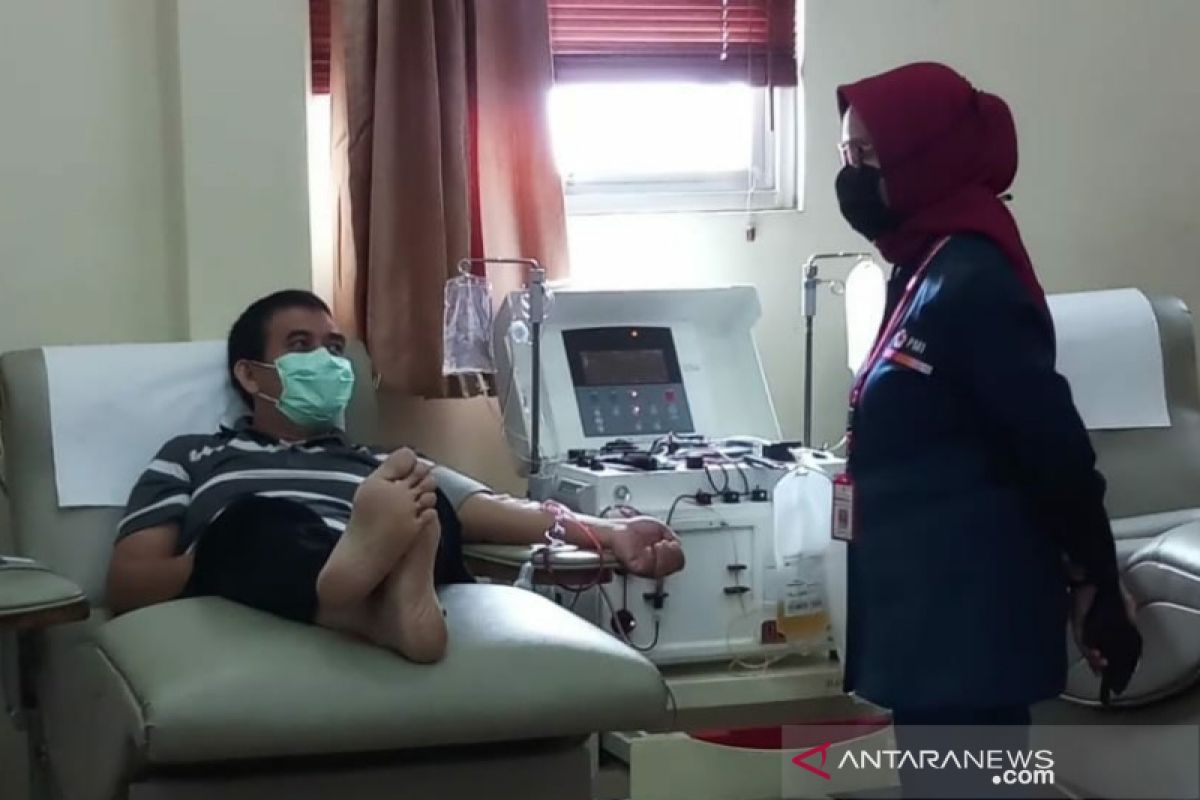
"We are ready to collect the blood plasma, yet we are currently awaiting official guidelines apart from a minimum requirement of two weeks after their symptoms resolve and only if they then test negative for COVID-19."Jakarta (ANTARA) - The Indonesian Red Cross for Jakarta region (PMI Jakarta) expressed willingness to collect blood of recovered COVID-19 patients for trial of treatment using convalescent blood plasma as proposed in Indonesia by Eijkman Molecular Biology Institute.
"We are ready to collect the blood plasma, yet we are currently awaiting official guidelines apart from a minimum requirement of two weeks after their symptoms resolve and only if they then test negative for COVID-19," Salimar Salim, head of the transfusions unit at PMI Jakarta, expounded on Friday.
Salim revealed that the organization was currently also forging cooperation with the hospitals since "they have the data of patients that had recovered from the disease."
PMI Jakarta owns some seven blood plasma collection devices, of which three can be utilized for this plan, as well as the facilities to separate plasma from blood using the plasmapheresis method.
The plasma will later be tested by Eijkman at some Biosafety Level 3 laboratories to ensure the effectiveness, safety, and security of COVID-19 patients. Related news: Red Cross working with Army to fight COVID-19
Related news: PMI to send 10 thousand N95 masks to Hong Kong
Translator: Livia Kristianti, Suwanti
Editor: Yuni Arisandy Sinaga
Copyright © ANTARA 2020