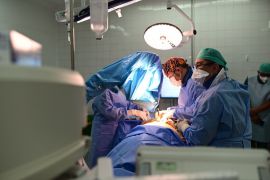
The stem cell clinical trial is an outcome of a collaboration between the Health Research and Development Agency of the Ministry of Health and the Daewoong Infion Korea Biopharmaceutical Company, according to a press release from the Ministry of Health received in Jakarta, Wednesday.
The cooperation agreement is an implementation of the Indonesia and South Korea Health Cooperation Agreement inked on November 9, 2017.
Collaboration on clinical trials of mesenchymal stem cell therapy was one of the agendas discussed at the virtual Joint Working Group between both nations on August 6, 2020, the second one ever to be held.
The health ministry of Indonesia and Korea also support collaboration between the government and health industry of both nations.
Collaboration is conducted to expedite the discovery of effective therapies for COVID-19 by conducting phase one clinical trials of MSC therapy.
Stem cell therapy, labeled “modern medicine,” is expected to address the ARDS ailment arising in COVID-19 patients.
The treatment does not work directly to kill viruses but rather functions as an immunomodulator that suppresses the production of reactive substances causing hyperinflammation and injuring lung tissue. Furthermore, mesenchymal stem cells have an anti-fibrotic effect that can replace fibrotic or injured lung tissue due to hyperinflammation.
According to outcomes of studies in other countries published in the form of systematic reviews and meta-analyzes, mesenchymal stem cells have been found to be a safe treatment, with marginal side-effects. They are also reportedly beneficial in lowering mortality and boosting lung function in patients suffering from ARDS.
Mesechymal stem cell products have been subject to the pre-clinical trial phase and proven to be safe and efficacious.
Acting Head of the Health Research and Development Agency, Dr Slamet, MHP, voiced his support for the preparation of this clinical trial.
The R&D Agency spots huge potential in the use of mesenchymal stem cells as a therapy in COVID-19 patients. Dr Slamet is optimistic that the collaboration would speed up the discovery of effective therapies for COVID-19 patients.
In the meantime, Head of the Research and Development Center for Resources and Health Services of the Ministry of Health's R&D Agency, Irmansyah, concurrently the research coordinator, noted that it was conducted at one of the referral hospitals of the Ministry of Health.
Irmansyah is optimistic of receiving support and cooperation of all parties involved in the implementation of research, right from the phase of preparation, down until the final stage of obtaining results and publications in reputable scientific journals.
Furthermore, the Ministry of Health welcomed the proposed collaboration with PT Daewoong Infion on clinical trials for COVID-19 drug candidates in which Daewoong had completed the phase one clinical trials in Korea and India.
Related news: Indigenous vaccine in the making
Related news: Indonesia currently developing its own Red and White vaccine: Jokowi
EDITED BY INE
Translator: Aditya Ramadhan/Aria Cindyara
Editor: Fardah Assegaf
Copyright © ANTARA 2020