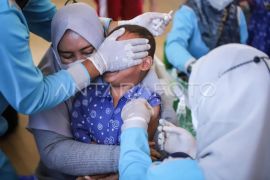
As many as 21 of the 248 volunteers have even received the second vaccine injections without having serious symptomsBandung, W Java (ANTARA) - The Padjadjaran University's Research Team for the Clinical Trials of Sinovac COVID-19 Vaccine revealed that 248 volunteers, including West Java Governor Ridwan Kamil, had been administered the vaccine.
"As many as 21 of the 248 volunteers have even received the second vaccine injections without having serious symptoms," the Unpad's research team manager, Eddy Fadliyana, said in a statement in Bandung, the capital of West Java Province, on Sunday.
None of the participating volunteers suffered serious symptoms after receiving the first and second vaccine injections, Fadliyana said, adding that the research on the clinical trials could be concluded successfully to enable the vaccine to be produced.
Among the volunteers who have received the vaccine injections are West Java Governor Ridwan Kamil and several officials of the provincial government.
Head of the Disease Prevention and Control Unit at Bandung City's Health Office Rosye Arosdiani Apip said earlier that the first batch of volunteers participating in the phase III clinical trials received the vaccine injections at several health centers.
The health facilities comprise Unpad's Health Center and Faculty of Medicine as well as the Garuda, Ciumbuleuit, Dago, and Sukapakir Public Health Centers.
In the first batch, one volunteer failed to participate in the clinical trial at the Garuda Public Health Center after receiving a reactive result in his rapid test, Apip revealed.
The Padjadjaran University's Research Team for Clinical Trials of Sinovac COVID-19 Vaccine need 1,620 volunteers who meet all required criteria for being able to participate in the clinical trials.
State-owned pharmaceutical holding company Bio Farma recently collaborated with Chinese company Sinovac to produce the COVID-19 vaccine for Indonesia. The COVID-19 vaccine material from Sinovac arrived in Indonesia on July 19.
Novel coronavirus infections initially surfaced in the Chinese city of Wuhan at the end of 2019.
Since then, COVID-19 has spread to over 215 countries and territories, including 34 provinces of Indonesia, with a massive spurt in death toll.
To tackle this COVID-19 pandemic, Indonesia is leaving no stone unturned to develop a vaccine to fight the virus.
Currently, in addition to the Sinovac COVID-19 vaccine, Indonesian scientists are working on a vaccine named after the country's national flag, Merah Putih (Red and White).
Related news: 15 mln Indonesians likely to get COVID-19 vaccine by year-end
Related news: Govt plans to distribute COVID-19 vaccines for free in 2021
Translator: Bagus AR, Rahmad Nasution
Editor: Fardah Assegaf
Copyright © ANTARA 2020