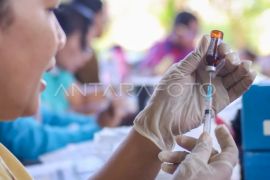
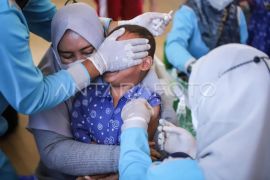
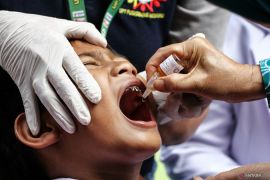
This vaccine is quite safe. It has no serious side effects on its usersJakarta (ANTARA) - President Joko Widodo has picked January 14 and January 15, 2021 to launch the initial stage of Indonesia’s mass vaccination program against COVID-19.
He has also decided to lead the immunization drive by receiving the country's first vaccine shot on January 13, 2021, in an event that will be broadcast live.
The Indonesian government will commence its vaccine program with President Widodo getting the first COVID-19 vaccine shot and the vaccination drive will continue on January 14 and 15 in several regions across the country, Home Affairs Minister Tito Karnavian announced on Tuesday.
The President volunteered to be the first to receive the vaccine to boost the public's confidence on its safety, Health Minister Budi Gunadi Sadikin explained.
The President will receive China's Sinovac vaccine a day or two after the Food and Drug Supervisory Agency (BPOM) issues an emergency-use authorization for the vaccine.
The Phase III clinical trials of the Sinovac vaccine are being conducted by the University of Padjajaran and state-owned pharmaceutical company Bio Farma in Bandung, West Java, since August, 2020.
BPOM said the results of the Phase III clinical trials of the Sinovac vaccine will come out in the first week of January, 2021. The BPOM will also obtain data on clinical trials of the vaccine conducted in Brazil and Turkey in order to make a decision.
The BPOM said it has discovered two types of data indicating the Sinovac vaccine's safety, based on the results of clinical trials in the past couple of months.
The two types of data pertain to the vaccine’s immunogenicity and efficacy, BPOM spokesperson for COVID-19 vaccination, Lucia Rizka Andalusia, noted in a written statement released on Tuesday.
"This vaccine is quite safe. It has no serious side effects on its users," Andalusia noted during an Alinea Forum on ‘Halal and Security of COVID-19 Vaccine’.
She revealed that the immunogenicity data showed a good immune response in the body resulting in antibody production.
The data may help dispel lingering doubts about the vaccine among the public, she stated.
Meanwhile, executive director of the Institute for Food, Drug, and Cosmetic Studies at the Indonesian Ulemas Council (LPPOM MUI), Muti Arintawati, said she is yet to ascertain whether the Sinovac vaccine can be considered halal as more information is awaited.
The LPPOM MUI's auditors have intensively studied raw materials for production of the vaccine through literature, journals, and expert information. Even if the information is complete, the MUI will still wait for BPOM's decision on the vaccine’s safety and efficacy to decide whether to issue a halal certificate or not, according to Arintawati.
So far, three million doses of the COVID-19 vaccine manufactured by China’s Sinovac Biotech have arrived in Indonesia in two batches — 1.2 million doses arrived on December 6, 2020, and 1.8 million doses on December 31, 2020. They have been distributed to various regions in Indonesia starting January 3, 2021.
The government has set a target to dispatch 29.55 million doses of the COVID-19 vaccine to the regions by March, 2021.
"Our target is that for January, 5.8 million (doses of) the vaccine must reach the regions. In February, 10.45 million (doses of the) vaccine must be distributed again to the regions, and thereafter, 13.3 million (doses of) vaccine must also be distributed in March," President Widodo noted at a closed-door meeting attended by ministers and governors through video-conferencing.
In the long term, at least 70 percent of the population, or 181.5 million people, are expected to be vaccinated to establish herd immunity.
For that purpose, the government is preparing to stockpile 329.5 million doses of COVID-19 vaccines from various manufacturers.
"I also need to mention about the number of vaccine doses that we have ordered. The firm orders include three million plus 122.5 million (doses) from Sinovac, then 50 million from Novavax, 54 million from Covax GAVI, 50 million from AstraZeneca, and 50 million (doses of the) vaccine from Pfizer. It means the total number of firm orders reaches 329.5 million (doses of) vaccines," Jokowi noted at the State Palace on Wednesday.
Novavax is being manufactured by the United States and Canada, AstraZeneca by the UK, and Pfizer is being jointly produced by pharma companies based in Germany and the United States. Covax GAVI is a multilateral cooperation between the World Health Organization (WHO) and the World Vaccine Alliance (GAVI) that comprises 171 countries, with a target of providing two billion vaccines by the end of 2021.
The Indonesian government has set aside a budget of about Rp73 trillion for procuring COVID-19 vaccines for the 2021 immunization program, Coordinating Minister for Economic Affairs, Airlangga Hartarto, said earlier.
The Health Ministry earlier said that the mass vaccination program against COVID-19 would take 15 months to complete, but Jokowi has asked the ministry to complete it in less than a year. The Health Ministry promised to work hard to meet the President's request.
The ministry has so far dispatched the first batch of 700 thousand doses of the Sinovac vaccine to the regions. In mid-January, raw materials for manufacturing 15 million doses of the COVID-19 vaccine are scheduled to arrive from China. They will be processed into vaccines by PT Biofarma, as part of a cooperation with Sinovac. The vaccines will thereafter be sent to different regions.
In a survey conducted by the Indonesian General Election Study (LKPI), 81.7 percent of the respondents have said they are ready to receive COVID-19 vaccine doses under the government’s immunization program.
The government will carry out the COVID-19 vaccination program in two phases, according to Health Minister Sadikin. The first phase will last from January to April, 2021, and the second from April, 2021 to March, 2022.
Around 1.6 million healthcare workers, 17.4 million public service officers, and 21.5 million elderly people will be prioritized in the first phase of the program.
In the second phase of the program, vaccinations will be provided to 63.9 million vulnerable people living in areas with a high risk of transmission and 77.4 million persons from other communities, using a cluster approach, depending on the availability of vaccines.
Sadikin said the government consulted the Indonesian Technical Advisory Group on Immunization (ITAGI), an independent agency, while mapping out the vaccination plan.
The President said he is optimistic that the transmission of the SARS-CoV-2 virus that causes COVID-19 would be brought under greater control after vaccination.
Jokowi, however, reminded citizens to adopt a disciplined approach in implementing the 3M health protocols to prevent the transmission of COVID-19 by wearing masks, maintaining distance, and washing hands.
"Do not go to crowded places and maintain your distance, as the key lies there until the vaccination is completed. Worldwide-vaccination is estimated to be competed in three and a half years," the President stated.
Despite the vaccination program, the authorities have advised Indonesians to continue applying the 3M health protocols.
"We must not become lax about 3M, not only in wearing masks, maintaining our distance, and washing our hands properly, and often. A crowd could become a super spreader. It is of no use for us to conduct tracing and it's pointless if there is still a crowd," Karnavian remarked.
Chairperson of the Task Force for COVID-19 Handling, Doni Monardo, also called for consistent public discipline in implementing health protocols.
"The narrative about this vaccine should not make people think that after the vaccine is available, everything will be over. No, the vaccine will not immediately stop COVID-19. The vaccine will not help unvaccinated people to escape COVID-19," he stated.
Related news: Red-and-White vaccine development 60 percent complete: Eijkman
Related news: President to be vaccinated at least 48 hours after authorization
Related news: Vaccine drive would push economic growth to 5%: Hartarto
Editor: Gusti Nur Cahya Aryani
Copyright © ANTARA 2021