"Studies by the World Health Organization (WHO) have shown that COVID-19 vaccination can reduce the incidence of serious illness, hospital admission, and mortality in the elderly," ITAGI chairwoman Professor Dr. Sri Rezeki Hadinegoro said at a webinar on “The Journey of COVID-19 Vaccination: The Importance of Booster Vaccinations in a Pandemic Period,” on Saturday.
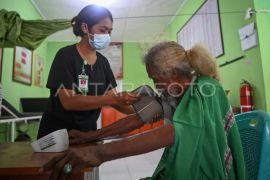
The COVID-19 booster can be administered to people aged 60 and up after an interval of at least three months once they complete the primary vaccination (first and second doses), she added.
Booster vaccinations can be homologous or heterologous, with vaccine regimens available in the field that have obtained an emergency-use permit from the National Agency of Drug and Food Control (BPOM) and are in accordance with ITAGI recommendations, Hadinegoro said.
A homologous booster vaccination uses the same vaccine as the primary vaccine platform type in the booster. For example, if the AstraZeneca vaccine is administered for the first and second dose, then it is also provided as a booster.
Meanwhile, if a booster is heterologous, recipients get a different booster vaccine than the primary vaccine. For example, if they get Sinovac for the first and second dose, they may get AstraZeneca for the booster dose.
A booster vaccine is considered important for dealing with the coronavirus, which has continued to mutate. Health studies on the COVID-19 vaccine have shown that antibodies developed through primary vaccination decline within six months, Hadinegoro pointed out.
"This booster can increase antibodies, very significantly," she said.
In addition to the elderly, other groups who need to get a booster shot are people who have comorbid diseases and people who have immune deficiencies.
Related news: COVID-19 vaccination: First dose coverage reaches 201.24 mln
In people with comorbid diseases, the vaccine can be given as long as the disease is controlled. For example, a booster vaccine can be given to people with diabetes as long as there are no acute complications. It is advisable for people to consult a doctor before vaccination if they have a comorbidity.
Hadinegoro noted that around 76 percent of Indonesian people have not received the booster vaccination. Data on adverse events following immunization (KIPI) has shown that the viral vector COVID-19 vaccine is safe to use for both primary and booster vaccinations.
"Until now, KIPI surveillance data shows that the viral vector COVID-19 vaccine is safe as a primary or booster (vaccine)," Dr. Hinky Hindra Irawan Satari said at the same webinar.
Reports submitted to KIPI National Committee have stated that the side effects of vaccination are mild and curable, such as fever, nausea, and aches.
Related news: Booster requirement for travel if COVID-19 keep increasing: Minister
Translator: Natisha Andarningtyas, Resinta
Editor: Rahmad Nasution
Copyright © ANTARA 2022