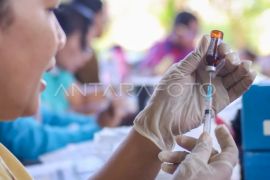
The ministry's Communication and Public Service Bureau on Saturday stated that the TB vaccine could offer economically affordable protection and maintain the sustainability of community productivity.
"If we want to achieve TB elimination by 2030, we only have three years to develop the TB vaccine to be used in 2028. We must focus on development of the TB vaccine," the minister stated as a board member at the meeting.
The 37th STP Board Meeting, which took place on Friday (February 9), was a forum for collaboration and communication for organizations and individuals from various sectors that are committed to eradicating Tuberculosis.
Indonesia, as a country that is also affected by TB, has convinced all G20 member countries to make adequate investments for availability of the TB vaccine in the next three years, Sadikin remarked.
According to Sadikin, the TB vaccine currently available in the country is the Bacillus Calmette-Guerin (BCG) vaccine as partial protection to prevent serious TB in babies and toddlers, but it does not provide enough protection to children and adults from TB.
Hence, he stated that the development of an effective TB vaccine should cover all age groups, especially for children and adults, with the ability to reduce incidence by 90 percent and lower mortality by 95 percent.
Related news: TB vaccine ready for third phase clinical trials: minister
TB vaccine candidates should also be able to overcome drug resistance, wherein bacteria fail to respond to standard treatments that are generally effective for treating TB infections, he pointed out.
Currently, several TB vaccine candidates under development already have the ability to prevent TB in children and adults, replace or strengthen the BCG vaccine, prevent recurrence in patients who have completed treatment, or shorten the duration of treatment, Sadikin noted.
He also stated that Indonesia is actively contributing to three clinical trials of TB vaccine candidates. The first is the vaccine developed by the Bill & Melinda Gates Foundation (BMGF). This vaccine, which was originally developed by the British pharmaceutical company, GSK, uses recombinant protein.
Second, a vaccine developed in collaboration with a pharmaceutical company from China, CanSinoBio, and a biopharmaceutical company from Indonesia, Etana. The development of this vaccine uses a viral vector and is currently in the first phase of clinical trials.
Lastly, a vaccine developed by a biotechnology company from Germany, BioNTech, and a pharmaceutical company from Indonesia, Biofarma. The development of this vaccine uses mRNA technology and is currently going to phase two of clinical trial in Indonesia.
"With this investment, we do not only save lives but also increase economic growth in the long-term," the minister remarked.
Related news: Support WHO initiative to develop new TB vaccine: Indonesia
Translator: Andi Firdaus, Resinta Sulistiyandari
Editor: Yuni Arisandy Sinaga
Copyright © ANTARA 2024